I. Industry Background
Pharmaceutical manufacturing processes generate substantial quantities of dust, including active pharmaceutical ingredients (APIs), excipients, and fine particulates. This dust is characterized by its fine particle size, high dispersibility, hygroscopicity, and—in some instances—toxic or irritant properties. If left uncontrolled, such dust not only contaminates the production environment but also compromises product quality and poses significant health risks to operating personnel.
The pharmaceutical industry imposes stringent requirements on dust removal equipment:
High-efficiency dust collection to ensure clean workshop air;
Prevention of cross-contamination to ensure compliance with GMP standards;
Resistance to moisture and caking to guarantee stable, long-term equipment operation;
Safety safeguards to prevent the inhalation of toxic dust.
Consequently, pulse bag dust collectors have emerged as a core component of dust control systems within the pharmaceutical sector, finding widespread application in processes such as powder drying, granulation, packaging, and material conveying.
II. Characteristics of Pharmaceutical Dust
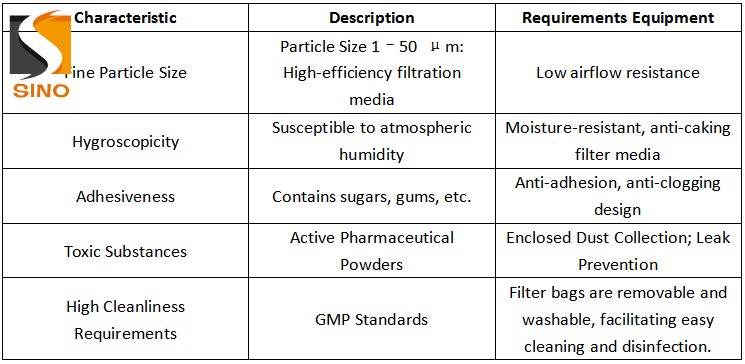
It is evident, therefore, that dust removal systems in the pharmaceutical industry must simultaneously incorporate features such as highly efficient dust collection, hygiene and cleanliness, moisture and clogging prevention, and sealed containment.
III. Application Advantages of Pulse Bag Dust Collectors
1. High-Efficiency Dust Removal
Features air-box pulse jet or compartmentalized pulse jet cleaning, achieving a dust removal efficiency of ≥99.9%;
Filter media options include PTFE-laminated or washable polyester filter bags, effectively balancing the capture of fine particles with resistance to caking;
Emission concentrations can be controlled to ≤10 mg/Nm³, fully meeting the stringent cleanliness requirements of the pharmaceutical industry.
2. Moisture-Resistant and Anti-Adhesion Design
Filter bag surfaces utilize PTFE lamination or anti-stick filter media to minimize dust adhesion;
The dust hopper can be equipped with heating or thermal insulation devices to prevent dust from absorbing moisture and caking;
Features an automated pulse-jet cleaning cycle to ensure timely and effective removal of dust from the filter bag surfaces.
3. Enclosed Design and Cross-Contamination Prevention
Employs fully enclosed ductwork and dust collection hoods to prevent dust dispersion;
Filter bags are removable for cleaning or replacement, thereby ensuring compliance with GMP production environment standards;
The dust recovery system is seamlessly integrated with the production process to maximize raw material utilization.
4. Intelligent Control and Safety Assurance
A PLC system provides real-time monitoring of differential pressure, airflow volume, and temperature, automatically adjusting the pulse-jet cleaning cycle;
Integrated alarm systems provide protection against operational anomalies, safeguarding both equipment and personnel;
Supports remote monitoring capabilities, facilitating the centralized management of dust control systems across multiple workshops.
IV. Typical Application Stages
(1) Drying Section
The drying of pulverized medicinal powder generates a significant amount of fine dust;
A pulse-jet baghouse dust collector is installed at the drying outlet to provide localized point extraction combined with centralized dust collection;
Anti-caking filter media are utilized to ensure the continuous operation of the system.
(2) Granulation and Mixing Section
During the mixing of active pharmaceutical ingredients with excipients, secondary dust dispersion is pronounced;
The dust collector is hermetically connected to the mixer to ensure a clean working environment;
The anti-stick and anti-caking design of the filter bags guarantees stable system operation.
(3) Packaging and Conveying Section
Dust tends to disperse easily from packaging machines and conveying pipelines;
The pulse-jet baghouse dust collector employs localized point extraction to prevent dust spillage;
Collected dust can be recycled and reused, thereby reducing raw material waste.
V. Typical Project Case Studies
Case Study 1: Pharmaceutical Powder Collection in the Drying Section
Operating Conditions: Fine dust particles; highly hygroscopic
Solution: Stand-alone pulse-jet baghouse dust collector (48 bags) utilizing PTFE-membrane filter media
Results: Dust removal efficiency ≥99.9%;
Emission concentration ≤10 mg/Nm³;
Filter bags are removable for cleaning; system operates stably.
Case Study 2: Dust Control in the Granulation and Mixing Section
Operating Conditions: Active pharmaceutical ingredients containing excipients; highly adhesive dust
Solution: Compartmentalized pulse-jet baghouse dust collector utilizing anti-stick polyester filter media
Results: Dust removal efficiency ≥99.85%;
System operates continuously and stably; no dust dispersion observed;
Filter bags feature a long service life and allow for convenient maintenance.
VI. Key Points for Operation and Maintenance
Differential Pressure Monitoring: Ensure timely dust removal to prevent filter bag clogging;
Dust Hopper Design: Maintain an appropriate slope; apply heating/moisture-proofing measures when necessary;
Filter Bag Maintenance: Conduct periodic inspections, cleaning, or replacements to ensure compliance with GMP requirements;
Sealed Ductwork: Prevent dust leakage and safeguard the workshop environment;
Intelligent Control System: Automatically adjust pulse-jet cleaning cycles and trigger alerts for abnormal conditions;
Dust Recovery and Utilization: Minimize raw material waste and enhance economic efficiency.
VII. Conclusion
Dust in the pharmaceutical industry is characterized by its fine particle size, hygroscopic nature, and—in some cases—toxic or irritant properties; consequently, this sector places rigorous demands on dust removal equipment.
With its advantages in high-efficiency dust collection, resistance to caking and adhesion, intelligent control capabilities, and sealed containment, the pulse bag dust collector has emerged as the preferred equipment for dust control within the pharmaceutical industry.
Through scientific design, standardized operation, and intelligent management, this technology not only ensures compliance with environmental standards but also extends equipment service life, enhances production efficiency, and safeguards workshop safety and hygiene—thereby providing a robust guarantee for green pharmaceutical manufacturing.
Contact: Jane
Phone: +86-136 0202 1569
E-mail: info@sinobaghouse.com
Add: No.14 Building, Jingjin Shishang Plaza, Wuqing District, Tianjin, China